In November last year, the House of Representatives Standing Committee on Health, Aged Care and Sport delivered its bipartisan report “The New Frontier: Delivering better health for all Australians” on the approval processes for new drugs and novel medical technologies in Australia.
Australian Health Journal spoke to some of the industry bodies who were part of the Inquiry, for their comments on the process, the report and hopes in the recommendations being implemented.
Professor John Zalcberg OAM Australian Clinical Trials Alliance (ACTA) Board Director & former ACTA Board Chair
Shanny Dyer, Chief Executive Officer, ARCS Australia
Leanne Wells, Chief Executive Officer, Consumers Health Forum of Australia
Liz de Somer, Chief Executive Officer Medicines Australia
Ian Burgess, Chief Executive Officer,
Medical Technology Association of Australia
Watch Season 5, Australian Health Journal Episode S5E1
You Might also like
-
Continuing education program leads to better conversations
Medicines Australia’s Continuing Education Program (CEP) is designed to educate medical representatives to a recognised industry standard. Dr Tristan Ling, CEP Academic Lead and Project Manager at the College of Health and Medicine, University of Tasmania talks about the 900 students that come through the program each year.
The CEP is primarily directed at medical representatives working within the prescription medicines industry, but is also recommended to people who may not be currently employed within the industry but would like to pursue a career as a medical representative. It is also available to personnel working for organisations interacting with the pharmaceutical industry.
-
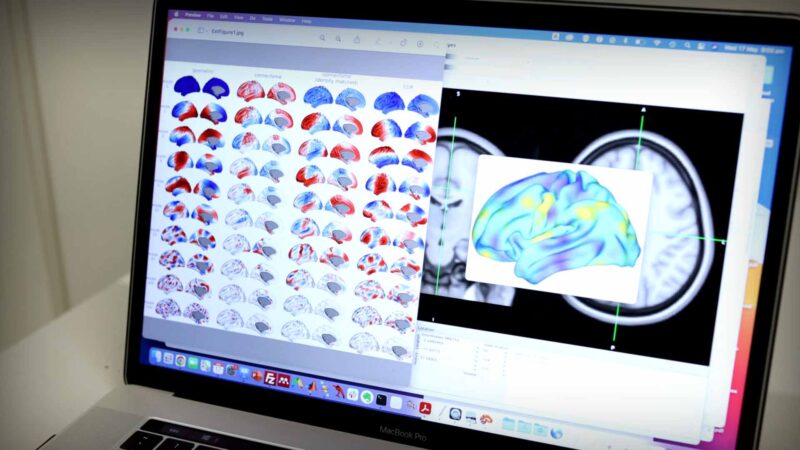
Landmark brain shape study
For over a century, researchers have thought that the patterns of brain activity that define our experiences, hopes and dreams are determined by how different brain regions communicate with each other through a complex web of trillions of cellular connections.
Now, a Monash University Turner Institute for Brain and Mental Health-led study has examined more than 10,000 different maps of human brain activity and found that the overall shape of a person’s brain exerts a far greater influence on how we think, feel and behave than its intricate neuronal connectivity.
-
Contrasting Population Needs and Alternative Funding Models
Dr Jaspreet Saini is a GP with a decade of experience, practicing in West Pennant Hills and Rooty Hill, New South Wales. He began his medical journey at Monash University, followed by internships and residency at Blacktown Hospital, in Western Sydney. Instead of confining himself to a single specialty, he pursued general practice to explore various facets of medicine.